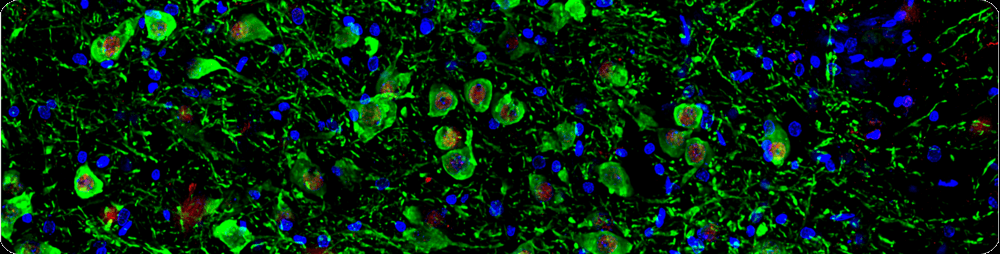
Differentiation of Induced Pluripotent Stem Cells (iPSCs) into Dopaminergic Neurons
Differentiating iPSCs into midbrain dopaminergic neurons offers incredible potential for researching neurological conditions, particularly Parkinson's disease. Whether you are investigating dopamine signaling pathways or modeling neurodegenerative diseases in vitro, this protocol outlines a clear, step-by-step method to efficiently drive your stem cells toward a dopaminergic fate. You will find it straightforward to follow, helping you achieve high-quality, functional neuronal cultures for all your downstream assays!
Featured Key Products
LDN193189 hydrochloride (LDN)
Potent, selective ALK2/ALK3 inhibitor which also promotes neural induction of hPSCs
View ProductRecombinant BDNF
Member of the neurotrophin growth factor family. Plays an important role in synaptic plasticity.
View ProductRecombinant human GDNF protein
GDNF is a key protein used to differentiate hPSC-derived neural progenitor cells into neurons
View ProductDAPT
γ-secretase inhibitor and classical notch inhibitor. Induces neuronal differentiation.
View ProductMaterials
- DAPT (HB3345)
- Recombinant human FGF-8a
- BDNF (HB3485)
- GDNF (HB5735)
- TGF-ß3
- Primary antibodies against dopaminergic markers such as tyrosine hydroxylase
- Secondary antibodies (see our range of validated secondary antibodies)
Media Recipes
Aseptically combine components and filter the media before use.
Media A - Day 1
| Component | Concentration |
|---|---|
| Knockout DMEM | 85% |
| Knockout Serum replacement | 15% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 mM |
| LDN193189 (HB5624) | 100 nM |
| SB431542 (HB3555) | 10 µM |
Media B - Days 2 - 3
| Component | Concentration |
|---|---|
| Knockout DMEM | 85% |
| Knockout Serum replacement | 15% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 mM |
| LDN193189 (HB5624) | 100 nM |
| SB431542 (HB3555) | 10 µM |
| SAG (HB3107) | 100 nM |
| Purmorphamine (HB3412) | 2 µM |
| Human FGF-8a | 100 ng/ml |
Media C - Days 4 - 5
| Component | Concentration |
|---|---|
| Knockout DMEM | 85% |
| Knockout Serum replacement | 15% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 mM |
| LDN193189 (HB5624) | 100 nM |
| SB431542 (HB3555) | 10 µM |
| SAG (HB3107) | 100 nM |
| Purmorphamine (HB3412) | 2 µM |
| Human FGF-8a | 100 ng/ml |
| CHIR99021 (HB1261) | 3 µM |
Media D - Days 6 - 7
| Component | Concentration |
|---|---|
| Knockout DMEM | 63.2% |
| Knockout Serum replacement | 11.6% |
| Neurobasal Medium | 24.8% |
| N-2 supplement | 0.13% |
| B-27 supplement | 0.26% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 µM |
| LDN193189 (HB5624) | 100 nM |
| CHIR99021 (HB1261) | 3 µM |
| SAG (HB3107) | 100 nM |
| Purmorphamine (HB3412) | 2 µM |
| Human FGF-8a | 100 ng/ml |
Media E - Days 8 - 9
| Component | Concentration |
|---|---|
| Knockout DMEM | 42.3% |
| Knockout Serum replacement | 7.6% |
| Neurobasal Medium | 49.3% |
| N-2 supplement | 0.26% |
| B-27 supplement | 0.5% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 µM |
| LDN193189 (HB5624) | 100 nM |
| CHIR99021 (HB1261) | 3 µM |
Media F - Days 10 - 11
| Component | Concentration |
|---|---|
| Knockout DMEM | 21% |
| Knockout Serum replacement | 3.8% |
| Neurobasal Medium | 74% |
| N-2 supplement | 0.38% |
| B-27 supplement | 0.74% |
| GlutaMax | 1x |
| MEM Non-Essential Amino Acids | 1x |
| 2-Mercaptoethanol | 10 µM |
| LDN193189 (HB5624) | 100 nM |
| CHIR99021 (HB1261) | 3 µM |
Media G - Days 12 - 13
| Component | Concentration |
|---|---|
| Neurobasal Medium | 96% |
| B-27 supplement | 1x |
| GlutaMax | 1x |
| DAPT (HB3345) | 10 µM |
| Dibutyryl cyclic-AMP sodium salt | 500 µM |
| Ascorbic acid (HB1238) | 200 µM |
| CHIR99021 (HB1261) | 3 µM |
| BDNF (HB3485) | 20 ng/ml |
| GDNF (HB5735) | 20 ng/ml |
| TGF-β3 | 1 ng/ml |